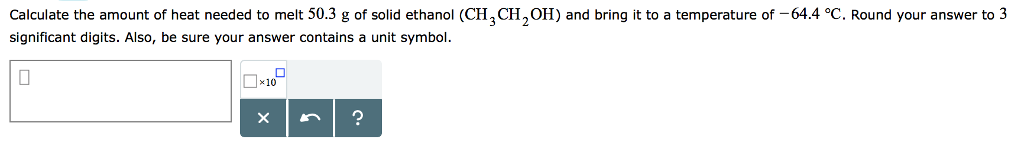
SOLVED: Suppose 250. mL flask is filled with 1.0 mol of 02 and 0.50 mol of NO The following reaction becomes possible: Nz (g)+02(g 72NO (g) The equilibrium constant K for this

SOLVED: Suppose 250. mL flask is filled with 1.0 mol of 02 and 0.50 mol of NO The following reaction becomes possible: Nz (g)+02(g 72NO (g) The equilibrium constant K for this

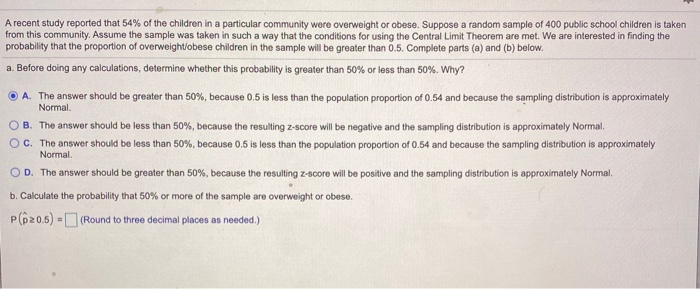
SOLVED: ateinar i dulu #h probability Sen nn E Hobabit pechabillty Jolate 'probal-luty nueunn probability Fedeti Colrulne five dog; shov Ruraty Two dogs 24. (5 points) Ehold birthday party for mny doz
![SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements](https://cdn.numerade.com/ask_previews/eb736f92-42a0-4b0d-8a1f-a15574a55833.gif)
SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements
![SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements](https://cdn.numerade.com/ask_previews/b2316b7e-8a0d-400d-8eac-eee10a259e93_large.jpg)
SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements
![SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements](https://cdn.numerade.com/ask_previews/9079e6d7-41c3-4bd1-b5ed-02793c816229.gif)
SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements

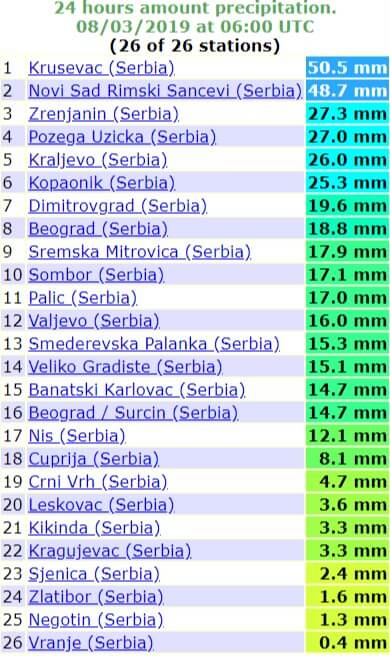
kolikšno luknjo predvrtati za vložek fid 50 od fischerja, Basic Math (Practicum) (KEE) | Quizlet - rhyanabbott.com
![SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements](https://cdn.numerade.com/ask_previews/42547854-3c39-4fb5-a702-aed8d18fff8e.gif)
SOLVED: Question 18 For - the reaction H2(g) 12(g) ZHI(g) , Kc = 50.2at 445 0C If [H2] = [I2] = [HI] 1.75 x 107-3) Mat 445 OC,which one of these statements